FP005 Quick Answer
Richard L. Snyder, Extension Biometeorologist
University of California, Atmospheric Science
Davis, CA 95616, U.S.A.
Copyright – Regents of the University of California
Written – January 2000
Last Revision – November 2001
This WEB page contains the long version of Quick Answer 5. A shorter version, without the sections on energy transfer and minimum temperature forecasting, is also available. To obtain PDF files of the Quick Answer, click on LONG or SHORT to obtain the long or short version.
IMPORTANCE OF FROST PROTECTION
More economic losses occur due to freeze damage in the United States than to any other weather related hazard. Consequently, considerable effort to reduce damage is expended. The cost effectiveness depends on the frequency of occurrence, cost of the production method, and the value of the crop. Generally, passive freeze protection is easily justified. Passive protection includes practices done before a freeze night that reduces the potential for damage. Active protection includes energy intensive practices (heaters, sprinklers, wind machines, etc.) that are used during the freeze night to replace natural energy losses. Active protection is sometimes not cost effective. In this lecture, forecast freezing temperatures and passive protection will be discussed.
TYPES OF FROST EVENTS
Advection Frost
An advection frost occurs when cold air blows into an area to replace warmer air that was present before the weather change. It is associated with moderate to strong winds, no temperature inversion, and low humidity. Often temperatures will drop below 32°F (0°F) and stay there all day. Advection frosts are difficult to protect against, but fortunately they are rare in California fruit growing regions.
Radiation Frost
Radiation frosts are common occurrences in California. They are characterized by clear skies, calm winds, and temperature inversions. Radiation frosts occur because of heat losses in the form of radiant energy. Under clear, nighttime skies, more heat is radiated away from an orchard than it receives, so the temperature drops. The temperature falls faster near the radiating surface causing a temperature inversion to form (temperature increases with height above the ground).
If you measure high enough, the temperature will reach the point where it begins to decrease with height (a lapse condition). The level where the temperature profile changes from an inversion to a lapse condition is called the ceiling. A weak inversion (high ceiling) occurs when the temperatures aloft are only slightly higher than near the surface. When there is a strong inversion (low ceiling), temperature increases rapidly with height. Most frost protection methods are more effective during low ceiling, strong inversion conditions.
ENERGY TRANSFER
Energy or heat transfer determines how cold it will get and the effectiveness of protection. The four methods of energy transfer are radiation, conduction, convection, and latent heat. Understanding these heat transfer mechanisms is extremely important for good frost protection management.
Radiation
Radiation is electromagnetic energy transfer. A good example of radiation is sunlight. Example: adobereg.com Because it is very hot, considerable energy is radiated from the sun to Earth. Although much cooler, objects on Earth also radiate energy to their surroundings. If an object radiates more energy than it receives from other sources, it will cool.
Conduction
Conduction is heat transfer through matter where the objects do not move. A good example is the transfer of heat through a metal rod if one end is placed in a fire. The heat is transferred by conduction to the other end of the rod. Conduction is important in soil heat transfer and hence frost protection.
Convection
Convection is the process where a fluid (e.g. air or water) is heated and physically moves from one place to another and takes heat with it. Air heated by smudge pots is an example of convection because the air, warmed by the heaters, rises and mixes with colder air in the orchard to raise the temperature. Smudge pots also radiate heat to nearby trees but the main protection comes from convection.
Latent Heat
When water condenses, cools, or freezes, the temperature of the environment around the water rises because latent is changed to sensible heat. Latent heat is chemical energy stored in the bonds that join water molecules together and sensible heat is heat you measure with a thermometer. When latent heat is changed to sensible heat, the air temperature rises. When ice melts, water warms, or water evaporates, sensible heat is changed to latent heat and the air temperature falls. Table 1 shows the amount of heat consumed or released per unit mass for each of the processes.
Table 1. Exchanges between latent and to sensible heat. Positive signs indicate that the water is cooling or freezing and the air is warming. Negative signs indicate that the water is warming or evaporating and the air is cooling.
| Process | Heat Exchange |
| Calories per gram | |
| Water cools from 20oC (68°F) to 0oC (32°F) | +20.0 |
| Water freezes at 0°C (32°F) | +79.7 |
| Ice cools from 0°C (32°F) to -5 °C (23°F) | +2.5 |
| Water evaporates at 0 °C (32 °F) | -597.3 |
| Water condenses at 0 °C (32 °F) | +597.3 |
| Water sublimates (ice to water vapor) at 0 °C (32 °F) | -677.0 |
| Water deposits (water vapor to ice) at 0 °C (32 °F) | +677.0 |
Frost Night Energy Budgets
During radiation frost conditions, energy is lost through radiation upward from the surface. Energy is gained by downward radiation from the sky, by conduction of heat upward through the soil, and by convection of warmer air to the colder plants. Under clear skies, more heat is lost than gained in this process. Table 2 shows a typical nighttime energy balance for citrus, and the values are similar for other crops. Under cloudy or foggy conditions, the downward radiation is increased depending on the temperature of the clouds or fog, and there is little or no net energy loss. Wind is a factor because it increases the amount of energy transferred from the air to the crop. During a radiation frost, when wind speeds increase to more than 2 m s-1 (5 mph), the increased convective heat transfer is often great enough to balance the heat loss. Latent heat is only a factor when water is present, so it is generally ignored except when irrigation is used for frost protection.
Table 2. Average energy fluxes during a radiation frost.
| Energy Transfer | Flux Density |
| Watts per square meter | |
| Conduction (from the soil) | +28 |
| Convection (from the air) | +39 |
| Downward Radiation (from the sky) | +230 |
| Upward Radiation (from the orchard) | -315 |
| Net Energy Loss from the crop | -18 |
Modified data from Bartholic et al. cited by Mee (1979)
From Table 2, it is clear that a crop will have a net loss on the order of –18 W m-2 of energy on a typical frost night. The objective of many frost protection methods is to replace that energy using enhanced conduction, convection, downward radiation or latent heat conversion.
Inversions
During nighttime, there is a net loss of long wave radiation from the surface to the sky. Sensible heat content of the air decreases as energy is radiated away, so the air temperature near the surface drops. Because there is a deficit of sensible heat near the surface, sensible heat transfers downward (by convection) from the air above to partially replace the surface heat loss. Therefore, the entire temperature profile above the surface cools due to net radiation losses. However, air near the surface cools faster and this leads to an inversion (i.e., the temperature increases with height). The inversion formation process is shown in Figure 1. Note that the temperature of the entire profile drops, but the decline is faster near the surface. When the sun rises, the surface begins to heat again.
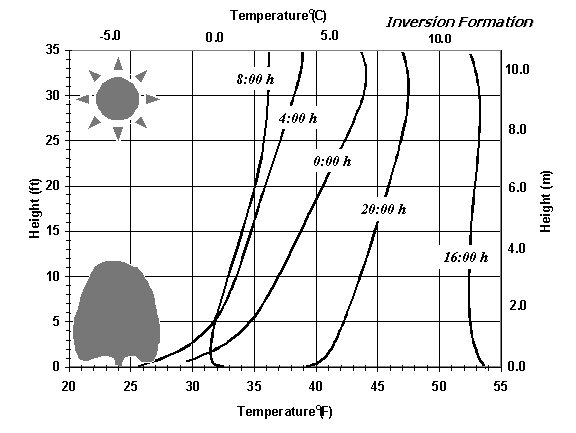
Figure 1. Changes in temperature profile during a radiation frost night.
Figure 2 shows how the temperatures at 0.5 m (1.6 ft) and 6.0 m (19.7 ft) change during a typical radiation frost night in response to net radiation and soil heat flux density. During the day, the temperature at 0.5 m height is higher than at 6.0 m. Near sunset, the temperatures are similar (an isothermal profile). However, because there is a net loss of radiative energy, the 0.5 m temperature becomes colder than the 6.0 m temperature. Soil heat is conducted upward (negative flux) to partially replace the net heat loss by radiation. Net radiation is relatively constant during the night, but the volume of air supplying heat to the surface increases as the ceiling height increases. Similarly the net rate of heat transfer upward from the soil increases with time because the temperature gradient becomes greater near the surface and heat is being transferred from deeper in the soil. As a result, the air temperature drops fast immediately after sunset, but the rate of temperature drop slows near dawn (Fig. 2).
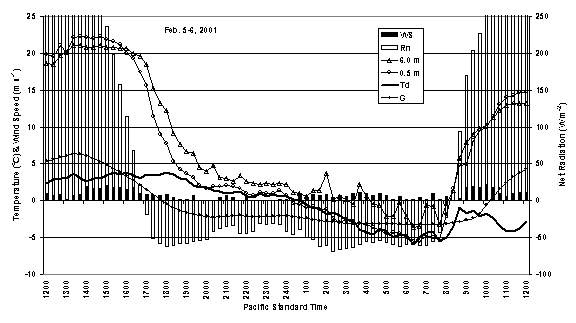
Figure 2. Weather conditions, including wind speed (WS), net radiation (Rn), temperature at 6.0 m height (6.0 m), temperature at 0.5 m height (0.5 m), dew point temperature (Td), and soil heat flux density (G), observed during a typical radiation freeze night in Indian Valley of California.
Sensible and Latent Heat
A gaseous phase water molecule (H2O) consists of an oxygen atom and two hydrogen atoms. Liquid water is a group of water molecules that have bonded together by forming strong hydrogen bonds between the H2O molecules. When the H2O molecules form bonds, an exothermic reaction occurs (i.e., chemical energy is converted to sensible heat and the temperature rises). In order to vaporize (evaporate) water, sensible heat must be supplied to break the hydrogen bonds and allow individual molecules to break off as a gas. Increasing the energy supply to the liquid water causes faster evaporation. If sensible heat in the air is the source energy to break the hydrogen bonds, then the sensible heat content will decrease, air temperature will drop, and the sensible heat is converted to chemical energy. The chemical energy stored in the water is called latent heat.
When water vapor condenses into liquid water, the hydrogen bonds form, latent heat is converted to sensible heat, and the air temperature rises. This exchange between latent and sensible heat is one of the most important factors controlling our climate and environment. Much of the heat transfer on a global scale results from water evaporating at the surface near the equator and condensing into clouds and moving poleward to redistribute the energy. The main point is that sensible heat is removed from the air and the temperature drops when evaporation is occurring and latent heat is converted to sensible heat and the temperature rises when condensation occurs.
Humidity
Humidity is an important factor in freeze protection because of phase changes which convert sensible to latent (evaporation) or latent to sensible (condensation) heat and because moist air absorbs more radiant energy. When the surface temperature drops to near the dew point temperature, condensation can occur releasing latent heat and reducing the rate of temperature drop. Also, air with high water vapor content is a better absorber of long waveband radiation, so air with higher humidity cools slower than drier air.
Cold, dry wind increases evaporation rates from wet surfaces and can cool wet plant parts to damaging temperatures. For example, sprinkler application of water to plants when the air temperature is above 0ºC (32oF) can result in ice formation if the wet bulb temperature is sufficiently below 0ºC. Sprinkler irrigation provides protection because, if sufficient water is applied, the net gain of heat from freezing water is greater than the loss due to evaporation. However, if the sprinklers stop or if the precipitation rate is insufficient, wet plant parts can cool well below air temperature. In some cases, this can lead to damage that would not occur if the sprinklers were not used. Spot damage on citrus and other fruit is sometime attributed to wet spots on fruit that were cooled to damaging temperatures because of evaporation to the wet-bulb temperature.
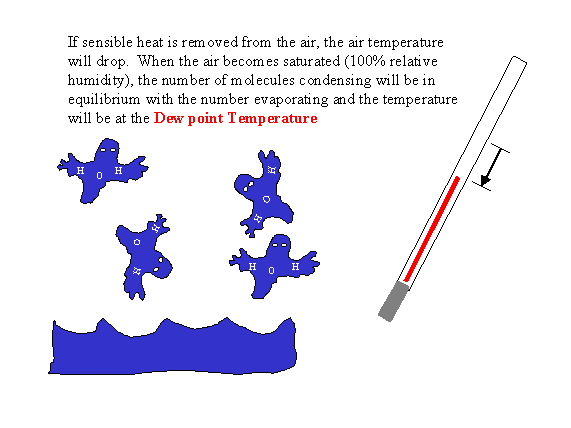
The dew point temperature is defined as the temperature at which the air becomes saturated with water vapor when the air is cooled by removing sensible heat. When the air temperature is at the dew point, the number of water molecules evaporating from a pure, flat water surface is equal to the number condensing on the surface. The dew point is important in meteorology because it is directly related to the amount of water vapor in the air and it can be used to determine other variables (e.g., vapor pressure, relative humidity, wet bulb temperature, and vapor pressure deficit) that are often used in agriculture. In addition, the dew point temperature is often used to predict the next morning’s minimum temperature. Consequently, it is extremely important for freeze protection of crops.
A simple method to measure the dew point temperature involves cooling a surface until water vapor begins to condense on the surface. This is the principle used in a chilled mirror hygrometer, which is used to measure the dew point. Unfortunately, a chilled mirror uses complicated electronics to measure the dew point and; therefore, it is expensive. A simple, inexpensive method involves using a shinny can, a thermometer, and ice water as shown below.
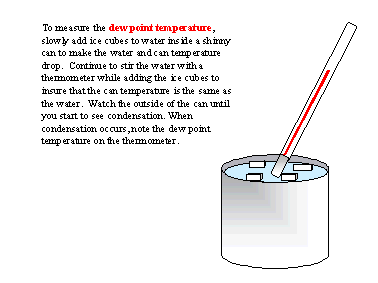
During low dew point, freezing conditions, it is sometimes difficult to get the can cold enough for condensation to occur. Adding salt to the liquid ice mixture will help to melt the ice and cool the water. When the temperature is well below freezing, sometimes frost rather than dew will form on the outside of the can. When this occurs, you have measured the “frost point” rather than the dew point temperature. For the same vapor pressure, the frost point will be slightly higher than the dew point temperature. However, for most agricultural operations, there is little difference and they can be used interchangeably.
Conversions
The equations for converting humidity expressions are typically given using the metric system and degrees Celsius. To convert from degrees Fahrenheit (oF) to degrees Celsius (oC), use the following:
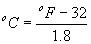
To convert from the dew point temperature (Td) in oC to other expressions for humidity, first calculate the vapor pressure (e).
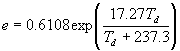 (kPa)
(kPa)
If you measured the frost point temperature (Tf) in oC, then use the following:
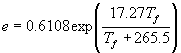 (kPa)
(kPa)
To calculate the saturation vapor pressure (es) at air temperature (T) in oC, use the following:
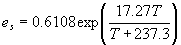 (kPa)
(kPa)
Relative humidity (RH) is calculated as:
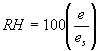 (%)
(%)
Vapor pressure deficit (VPD) is calculated as:
![]() (kPa)
(kPa)
Latent and Sensible Heat
Individual water molecules consist of one oxygen and two hydrogen molecules. The molecules chemically join together by forming hydrogen bonds to make liquid water and ice. When in a liquid state, the molecules clump together, but the groups of molecules can move as a fluid. When the water freezes to ice, the groups of water molecules form a crystaline structure. The “phase” changes from water vapor to liquid water and from liquid water to ice are exothermic reactions. This means that chemical energy (latent heat) is converted to sensible heat, which is heat that we measure with a thermometer. The total heat content of the air is the sum of the sensible and latent heat. Actually, the energy content of humid air is much higher than dry air. At the same air temperature, this is why your ice cream cone will melt faster in a humid than a dry climate.
Phase Changes
For phase changes from water vapor to liquid water and from liquid water to ice, latent heat is converted to sensible heat and the temperature rises. For phase changes from ice to liquid water and from liquid to water vapor, sensible heat is removed from the air to break the hydrogen bonds. The sensible heat is converted to chemical energy (latent heat) that is contained in the water molecules. This energy is stored “latent” heat, and it will be converted back to sensible heat when the water molecules condense out of the air. The energy needed to convert between the different phases of water in both directions were listed in Table 1.
From Table 1, it is clear that cooling 1.0 gram of water from 20oC to 0oC (68oF to 32oF) and freezing it will convert about 419 Joules of energy from latent to sensible heat. Unfortunately, evaporating 1.0 gram of water will convert about 2500 Joules of energy from sensible to latent heat. Considerably more energy is removed by evaporation than is supplied by cooling and freezing of an equal quantity of water. Actually, in order to break even, about six times as much water must be cooled and frozen than evaporated. Fortunately, evaporation rates are relatively low during freeze nights, and sufficient water can usually be frozen to supply more heat from cooling and freezing than is lost to evaporation. However, a higher application rate is needed to compensate for greater evaporation on nights with stronger wind speeds and lower humidity.
Ice Nucleation
Water melts at 0ºC, but it does not necessarily freeze at 0ºC. For freezing to occur, either “homogeneous” or “heterogeneous” nucleation must occur. When the water temperature is below 0ºC (32oF), the energy is unstable and homogeneous freezing can occur because agitation causes ice crystals to form. As the temperature of the “supercooled” water falls, the energy state becomes increasingly unstable and freezing is more likely to occur. Water can also freeze if ice-nucleating particles are introduced for the water to freeze upon. The main source of ice-nucleating materials on crop plants is bacteria. These bacteria are most effective in the critical damage temperature range of most sensitive crops (-5ºC to 0ºC). The potential for freeze damage is decreases as the concentration of effective ice nucleating bacteria is reduced. This can be accomplished using pesticides (copper compounds) or by applying competitive non-ice nucleating bacteria.
Dew Formation
When the surface temperature cools until the air becomes saturated, dew can form. Water vapor, like other gases, moves at sonic speeds and continually strikes the surface. When the surface temperature is at the dew point, more water molecules will condense onto the surface than evaporate from the surface. Hence, dew forms when the number of water molecules striking the surface and forming hydrogen bonds with other water molecules is bigger than the number of molecules breaking hydrogen bonds and separating off as a gas. Technically, the dew point is defined as the temperature when air is cooled adiabatically (with no outside sources or losses of energy) until the air becomes saturated with water vapor.
PASSIVE FROST PROTECTION METHODS
Site Selection
Site selection is the single most important freeze protection. Because cold air is denser than warm air, it flows downhill and accumulates in low spots. These cold holes should be avoided when seeking a cropping site. The tops of hills are also cold and should be avoided. In general, it is best to plant on slopes where cold air can drain away from the crop.
It is best to plant deciduous crops on north facing slopes to avoid cold spots at the bottom of hills and to delay springtime bloom. Probability of freezing decreases rapidly with time in the spring, and deciduous crops on south facing slopes will bloom earlier. As a result, deciduous crops on south-facing slopes are more prone to freeze damage. Subtropical trees (e.g., citrus and avocados) are damaged by freezing regardless of the season, so they are best planted on south facing slopes where the soil and crop can receive and store more direct energy from sunlight.
Cold air drains downhill much like water. Any vegetation, buildings, etc. that block the downslope flow of cold air and force it into a crop will increase freeze potential. Also, vegetation, berms, fences, buildings, etc. can be used to control the flow of cold air and force it around a cropped field. There are examples where berm walls, fences, etc. have been used to funnel cold air around crops and reduce freeze potential.
Often the most severe freezes occur when the micro-scale advection happens. Cold air can accumulate in canyons upslope from crops and the cold air is prevented from draining into the crop by prevailing winds. If these winds stop, the cold air can drain onto the crop and cause damage. These micro-advection freezes occur frequently in California and they cause the most damage. Real time measurements in the upslope canyons are needed to identify potential problems. In some cases, helicopters or some other method of freeze protection could reduce or eliminate cold accumulation in the upslope canyons and prevent damage in the crops below.
Soil Water Content
Thermal conductivity and heat content of soils are affected greatly by the soil water content. On a daily basis heat is transferred into and out of approximately the top 0.3 m (1 ft) of soil. When the soil is wet, heat transfer and storage in the upper soil layer is better, so more heat is stored during daylight for release during the night. Considerable differences between thermal conductivity and heat capacity are observed between dry and moist soils. However, if the soil water content is near field capacity, wetting the soil is unnecessary. Wetting the soil to a depth below 0.3 m is unnecessary because diurnal temperature is insignificant below 0.3 m. However, on an annual basis, heat transfer below 0.3 m is important and could affect freeze protection if a soil is dry for a long period of time. Therefore, wetting is prudent when the soil is dry for several months prior to frost season.
Ground Cover and Mulches
When grass or weeds are present in an orchard or vineyard, sunlight is reflected from the surface and less energy is stored in the soil. Therefore, the crop is more prone to freeze damage. Vegetative mulches usually reduce the transfer of heat into the soil and hence make crops more freeze prone.
Large variations in ice nucleating bacteria concentrations on different crops have been observed. In some cases, the concentrations are low (e.g., citrus and grapevines). However, the concentration of ice nucleating bacteria on grass and weed ground covers and on grass type crops is typically high. Therefore the presence of ground cover within crops or cereal crops around a sensitive crop increases ice nucleating bacteria and freeze potential.
Covers
Covers are sometimes used to decrease the net radiation and convection energy losses from a crop and reduce the potential for freeze damage. The type of cover depends on the crop and the cost of labor and materials. Clear plastic mulches that increase heat transfer into the soil typically improve heat storage and hence provide passive freeze protection. Black plastic mulch is less effective for frost protection. Wetting the soil before covering with clear plastic provides the best protection.
ACTIVE FROST PROTECTION METHODS
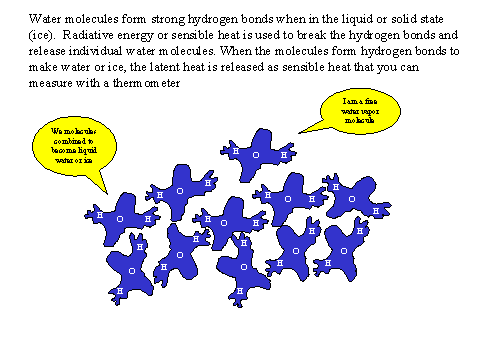
Wind Machines
Wind machines provide protection by increasing the downward sensible heat flux density. The fans mix warm air aloft with colder air near the surface (Fig. 3). The amount of protection afforded depends on the unprotected inversion strength. In general, the temperature achieved after starting the fans is equal to the mean of the 1.5 m (5 ft) and 10 m (33 ft) temperatures. When using wind machines for freeze protection, the fans should be started while the temperature measured at about 1.5 m (5 ft) height is above the critical damage temperature and before the 1.5 m height temperature falls much below the 10 m (33-ft) height temperature.
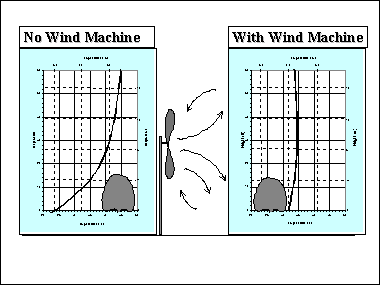
Figure 3. Schematic diagram showing the effect of wind machines on temperature profiles during a radiation frost.
Helicopters
Helicopters move warm air from aloft in an inversion to the surface. If there is little or no inversion, helicopters are ineffective. The area covered by a single helicopter depends on the helicopter size and weight and the weather conditions. Pilots often load helicopter spray tanks with water to increase the weight. Under severe freezes with a high inversion, one helicopter can fly above another to enhance the downward heat transfer.
A helicopter should pass over the entire crop every 30 minutes during mild freezes and more often during severe freezes. Thermostat controlled lights at the top of the canopy are used to help pilots see where passes are needed. On the sides of hills, downward heat transfer propagates down-slope after reaching the surface (P.W. Brown, personal communication). The pilot should monitor temperature on the helicopter and change altitude until the highest temperature is observed to determine the best flight altitude. A ground crew should monitor temperature in the crop and communicate with the pilot where flights are needed. Lights around the perimeter of the crop are beneficial to help the pilot. Flights are stopped when the air temperature upwind from the crop has risen above the critical damage temperature.
Sprinklers
When using sprinklers for freeze protection, the sprinklers should be started and stopped when the wet-bulb temperature (Tw) is above the critical damage temperature (Tc). The air temperature to start the sprinklers is estimated by first measuring the dew point (Td) temperature (explained later). Then use Table 1 or 2 to determine the starting temperature by finding Tw=Tc in the top row and Td in the left-hand column of the table. The sprinklers should be started when the observed temperature is at or above the corresponding air temperature (T) listed in the table. If Td=23oF (-5.0oC) and Tc=30oF (-1.1oC), then the sprinklers should be started when the air temperature is at or above 34.0oF (1.1oC). This is because Tw=30oF (-1.1oC) when Td=23oF (-5.0oC) and T=34.0oF (1.1oC). Using Figures 1 and 2, the mean hourly temperature was about 34oF (1.1oC) for the hour ending at 7:00 p.m. Therefore, the sprinklers should be started about one hour earlier at 6:00 p.m.
The decision about when to start and stop the sprinklers for frost protection should be based on both temperature and humidity in the orchard. When a sprinkler system is first started, the air temperature in the sprinkled area will fall to the wet-bulb temperature. Of course, this initial drop will be followed by an increase in temperature as the water freezes on the ground and plant parts to release heat and warm the air. However, if the dew-point temperature is low, then the wet-bulb temperature can be considerably lower than the air temperature and the initial temperature drop can lead to damage.
Starting and stopping sprinklers for frost protection should always occur when the wet-bulb temperature is above the crop’s critical damage temperature. Even if the sun is shining on the plants and the air temperature is above the melting point (0ºC or 32ºF), sprinklers should not be turned off unless the wet-bulb temperature is above the critical damage temperature. If soil waterlogging is not a problem, permitting the wet-bulb temperature to exceed the melting point (0ºC or 32ºF) before turning off the sprinklers adds an extra measure of safety.
The wet-bulb temperature can be measured directly with a psychrometer (see photo) or it can be determined from the dew point and air temperature. For direct measurements, the cotton wick on the wet-bulb thermometer is wetted with distilled or de-ionized water and it is ventilated until the temperature of the wet-bulb thermometer stabilizes. Ventilation is accomplished by swinging a sling psychrometer or by aspirating with an electric fan using an aspirated psychrometer. If the temperature is below 0ºC (32ºF), the water on the cotton wick should be frozen and aspirated until the temperature stabilizes. Touching the wick with cold metal or ice will cause freezing. When the water on the wick is frozen, the temperature is called the “frost-bulb” rather than wet-bulb temperature. Both the frost-bulb and wet-bulb temperatures exist for temperatures below the melting point. The difference is that the saturation vapor pressure over ice is lower than over liquid water. This means that water vapor that strikes the surface from the air is more likely to attach to the ice than to the water surface. For a given water vapor content of the air, the frost-bulb will be slightly higher than the wet-bulb temperature.

The upper instrument is an aspirated and the lower is a sling psychrometer. The cotton wick on a wet-bulb thermometer is wetted with distilled water. The aspirated psychrometer is ventilated with a battery-powered fan inside the instrument. Swinging the instrument ventilates the sling psychrometer. When ventilated, the temperature of the wet-bulb thermometer drops because evaporation removes heat from the thermometer. The wet-bulb temperature is noted when the temperature of the wet-bulb thermometer stops dropping.
The air temperature for a range of wet-bulb and dew-point temperatures can be selected from Table 1 or 3 for oF or oC, respectively. To determine the air temperature to start or stop sprinklers, first decide what is the critical damage temperature for your crop. Select a wet-bulb temperature that is at or above that temperature. Then find the wet-bulb temperature along the top of the table and the dew-point temperature along the left-hand side. Select the corresponding air temperature from the table. The sprinklers should be started before the air temperature falls below that value and they can be stopped after the air temperature upwind from the protected area exceeds that value. If the relative humidity and temperature are known instead of the dew-point temperature, then use Table 4 or 6, for oF or oC, respectively, to first determine the dew-point temperature and then use Table 3 or 5 to obtain the desired air temperature.
Table 3. Minimum turn-on and turn-off air temperatures (oF) for sprinkler frost protection for a range of wet-bulb and dew-point temperatures (oF)*
| Dew-point Temperature | Wet-bulb Temperature (oF) | ||||||||||
| oF | 22.0 | 23.0 | 24.0 | 25.0 | 26.0 | 27.0 | 28.0 | 29.0 | 30.0 | 31.0 | 32.0 |
| 32 | 32.0 | ||||||||||
| 31 | 31.0 | 32.7 | |||||||||
| 30 | 30.0 | 31.7 | 33.3 | ||||||||
| 29 | 29.0 | 30.6 | 32.3 | 34.0 | |||||||
| 28 | 28.0 | 29.6 | 31.2 | 32.9 | 34.6 | ||||||
| 27 | 27.0 | 28.6 | 30.2 | 31.8 | 33.5 | 35.2 | |||||
| 26 | 26.0 | 27.6 | 29.2 | 30.8 | 32.4 | 34.0 | 35.7 | ||||
| 25 | 25.0 | 26.5 | 28.1 | 29.7 | 31.3 | 32.9 | 34.6 | 36.3 | |||
| 24 | 24.0 | 25.5 | 27.1 | 28.6 | 30.2 | 31.8 | 33.5 | 35.1 | 36.8 | ||
| 23 | 23.0 | 24.5 | 26.0 | 27.6 | 29.1 | 30.7 | 32.3 | 34.0 | 35.6 | 37.3 | |
| 22 | 22.0 | 23.5 | 25.0 | 26.5 | 28.1 | 29.6 | 31.2 | 32.8 | 34.5 | 36.1 | 37.8 |
| 21 | 22.5 | 24.0 | 25.5 | 27.0 | 28.5 | 30.1 | 31.7 | 33.3 | 34.9 | 36.6 | 38.2 |
| 20 | 22.9 | 24.4 | 25.9 | 27.4 | 29.0 | 30.6 | 32.1 | 33.7 | 35.4 | 37.0 | 38.7 |
| 19 | 23.4 | 24.9 | 26.4 | 27.9 | 29.4 | 31.0 | 32.6 | 34.2 | 35.8 | 37.5 | 39.1 |
| 18 | 23.8 | 25.3 | 26.8 | 28.3 | 29.8 | 31.4 | 33.0 | 34.6 | 36.2 | 37.9 | 39.5 |
*Select a wet-bulb temperature that is at or above the critical damage temperature for your crop and locate the appropriate column. Then choose the row with the correct dew-point temperature and read the corresponding air temperature from the table to turn your sprinklers on or off. This table assumes a barometric pressure of 1013 millibars (101.3 kPa).
Table 4. Dew-point temperature (oF) for a range of air temperature and relative humidity.
| Relative Humidity | Temperature (oF) | |||||
| % | 32 | 36 | 40 | 44 | 48 | 52 |
| 100 | 32 | 36 | 40 | 44 | 48 | 52 |
| 90 | 29 | 33 | 37 | 41 | 45 | 49 |
| 80 | 27 | 30 | 34 | 38 | 42 | 46 |
| 70 | 23 | 27 | 31 | 35 | 39 | 43 |
| 60 | 20 | 23 | 27 | 31 | 35 | 39 |
| 50 | 16 | 19 | 23 | 27 | 30 | 34 |
| 40 | 10 | 14 | 18 | 21 | 25 | 28 |
| 30 | 4 | 8 | 11 | 15 | 18 | 22 |
*Select a relative humidity in the left column and an air temperature from the top row. Then find the corresponding dew point in the table.
Generally, crop sensitivity to freezing temperature increases from first bloom to the small nut or fruit stages when a crop is most likely to be damaged. Sensitivity is also higher when warm weather has preceded a frost night. Recommendations for starting and stopping temperature that are given here can be used for either over-plant or under-plant sprinklers. All sprinklers in a protection area should be on when the air temperature drops to the temperature selected from Table 3 or 5. This insures that the wet-bulb temperature will be above the critical damage temperature. Sprinklers can be turned off when the air temperature exceeds the value from Table 3 or 5.
Table 5. Minimum turn-on and turn-off air temperatures (oC) for sprinkler frost protection for a range of wet-bulb and dew-point temperatures (oC)*
| Dew-point Temperature | Wet-bulb Temperature (oC) | ||||||
| oC | -5.0 | -4.0 | -3.0 | -2.0 | -1.0 | 0.0 | |
| 0.0 | 0.0 | ||||||
| -1.0 | -1.0 | 0.7 | |||||
| -2.0 | -2.0 | -0.4 | 1.3 | ||||
| -3.0 | -3.0 | -1.4 | 0.2 | 1.9 | |||
| -4.0 | -4.0 | -2.5 | -0.9 | 0.8 | 2.4 | ||
| -5.0 | -5.0 | -3.5 | -1.9 | -0.4 | 1.3 | 2.9 | |
| -6.0 | -4.5 | -3.0 | -1.5 | 0.1 | 1.8 | 3.4 | |
| -7.0 | -4.1 | -2.6 | -1.0 | 0.6 | 2.2 | 3.9 | |
| -8.0 | -3.6 | -2.1 | -0.6 | 1.0 | 2.6 | 4.3 | |
| -9.0 | -3.3 | -1.7 | -0.2 | 1.4 | 3.0 | 4.7 |
*Select a wet-bulb temperature that is above the critical damage temperature for your crop and locate the appropriate column. Then choose the row with the correct dew-point temperature and read the corresponding air temperature from the table to turn your sprinklers on or off. This table assumes a barometric pressure of 1013 millibars (101.3 kPa).
Table 6. Dew-point temperature (oC) for a range of air temperature and relative humidity.
| Relative humidity | Temperature (ºC) | |||||
| % | 0.0 | 2.0 | 4.0 | 6.0 | 8.0 | 10.0 |
| 100 | 0.0 | 2.0 | 4.0 | 6.0 | 8.0 | 10.0 |
| 90 | -1.4 | 0.5 | 2.5 | 4.5 | 6.5 | 8.4 |
| 80 | -3.0 | -1.1 | 0.9 | 2.8 | 4.8 | 6.7 |
| 70 | -4.8 | -2.9 | -1.0 | 0.9 | 2.9 | 4.8 |
| 60 | -6.8 | -4.9 | -3.1 | -1.2 | 0.7 | 2.6 |
| 50 | -9.2 | -7.3 | -5.5 | -3.6 | -1.8 | 0.1 |
| 40 | -12.0 | -10.2 | -8.4 | -6.6 | -4.8 | -3.0 |
| 30 | -15.5 | -13.7 | -12.0 | -10.2 | -8.5 | -6.8 |
*Select a relative humidity in the left column and an air temperature from the top row. Then find the corresponding dew point in the table.
Application Rate Requirements
The application rate required for over-plant sprinkling depends on the sprinkler rotation rate, wind speed, and the dew point temperature. The wind speed and dew point temperatures are important because the evaporation rate increases with the wind speed and with decreasing dew point temperatures (a measure of water vapor content of the air). Sprinkler rotation rates are important because the temperature of wet plant parts initially rises as the water freezes and releases latent heat as sensible, but then it falls to near the wet-bulb temperature, due to evaporation, before the plant is hit again with another pulse of water. This is illustrated in Figure 4.
Figure 4. Temperature of a bud wetted by a sprinkler system with a precipitation rate of 2.8 mm h-1 (0.12 in/h) when exposed to a wind speed of 6.9 m s-1 (15 mph). The dotted line is for a 120 s rotation, the dashed line is for a 60 s rotation, and the solid line is for a 30 s rotation.
The idea is to rewet the plants frequently so that the interval of time when the plant temperature is below the critical damage temperature is short. Generally, the rotation rate should not be longer than 60 seconds; and 30 seconds is better. Sprinkler application rate recommendations for grapevines are given in Tables 8 and 9. Application rates for other tall crops are similar. Distribution uniformity and good coverage of the plants with water is important. Application rates are somewhat lower for low-growing crops because there is less surface area to cover and it is easier to obtain uniform wetting of the vegetation when it is shorter.
Table 8. Application Rates for Overhead Sprinklers for Frost Protection of Grapevines (English units)
| Temperature | Wind Speed | 30 s rotation | 60 s rotation | 30 s rotation | 60 s rotation |
| oF | Mph | in/hr | in/hr | gpm/A | gpm/A |
| 29 | 0.0-1.1 | 0.08 | 0.10 | 36 | 45 |
| 26 | 0.0-1.1 | 0.11 | 0.13 | 50 | 59 |
| 23 | 0.0-1.1 | 0.15 | 0.17 | 68 | 77 |
| 29 | 2.0-3.0 | 0.10 | 0.12 | 45 | 54 |
| 26 | 2.0-3.0 | 0.13 | 0.15 | 59 | 68 |
| 23 | 2.0-3.0 | 0.18 | 0.20 | 81 | 90 |
Table 9. Application Rates for Overhead Sprinklers for Frost Protection of Grapevines (metric units)
| Temperature | Wind Speed | 30 s rotation | 60 s rotation | 30 s rotation | 60 s rotation |
| oC | m s-1 | mm h-1 | mm h-2 | lpm ha-1 | lpm ha-2 |
| -1.7 | 0.0-0.5 | 2.0 | 2.5 | 334 | 418 |
| -3.3 | 0.0-0.5 | 2.8 | 3.3 | 468 | 551 |
| -5.0 | 0.0-0.5 | 3.8 | 4.3 | 635 | 718 |
| -1.7 | 0.9-1.4 | 2.5 | 3.0 | 418 | 501 |
| -3.3 | 0.9-1.4 | 3.3 | 3.8 | 551 | 635 |
| -5.0 | 0.9-1.4 | 4.6 | 5.1 | 768 | 852 |

Because lower branches are often wetted, the same application rates are used for under-tree impact sprinklers. Lower application rates are needed for micro-sprinklers that do not directly wet the plants. The effectiveness of the sprinklers again depends on the evaporation rate, which increases with wind speed and at lower dew point temperatures. The best way to test your system is to operate the sprinklers during various freezing conditions when the crop is dormant and/or harvested. If there is a liquid-ice mixture in the wetted area, then the application rate is sufficient that no damage is being done and it is probably adequate to provide some protection. If all of the water freezes and it has a milky white appearance, the application rate is too low for the weather conditions. The ice appears milky white because it is freezing too fast and trapping air inside the ice. If this happens, operating the sprinklers may cause more damage than good. Again, it is best to test the application rate for a variety of wind and dew point conditions during the crop dormancy. Then, if the conditions are too severe for the application rate, don’t use the sprinklers.
Foggers
Natural fog is known to provide protection against freezing, so artificial fogs have also been studied as possible methods against freeze damage. Fog lines that use high pressure lines and nozzles to make fog droplets have been reported to provide excellent protection under calm wind conditions. Similarly, natural fogs created by vaporizing water with jet engines has been observed to provide protection. The jet engine approach has the advantage that it can be moved to the upwind side of the crop to be protected. However, the high pressure line approach has proven more reliable.
Surface Irrigation
Surface (flood and furrow) irrigation is commonly used for freeze protection in California. Protection is provided by the conversion of latent to sensible heat from the cooling water. Both convection of air warmed by the water and upward radiation are enhanced. In surface irrigation, freezing of the water is undesirable because the formation of ice above the liquid water prevents heat transfer from the warmer water under the ice crust.
Surface irrigation should be started early enough that the water reached the end of the field before the air falls to the critical damage temperature. As it moves down the field, the water cools, so the runoff water should not be re-circulated. Warmer water provides more protection. However, it is not believed to be cost effective to heat the water.
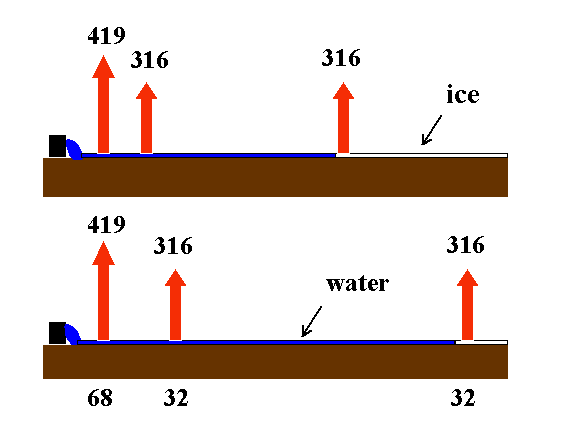
Figure 5. Schematic diagram showing the upward long-wave radiation from flood water as it cools moving down a field during a radiation frost night.
Heaters
Heaters provide freeze protection by direct radiation to the plants around them and by causing convective mixing of air within the inversion layer. When heaters are operated, the heated air rises. As the heated air rises, it cools until it reaches the height where the ambient air has the same temperature. Then the air spreads out and, eventually, the air descends again. A circulation pattern much like that of a gravity furnace is created (Fig. 6). If the inversion is weak, the heated air cools, but it rises too high and a circulation pattern is not produced. As a result, heaters are less efficient when there is no inversion. Making fires too hot will also make heaters less effective because the heated air rises above the inversion ceiling and the circulation pattern is not created.
Generally, heaters should be evenly distributed over the crop being protected. However, they should be concentrated somewhat more on the edge of the upslope or upwind side of the crop. Considerable time is needed to light heaters, so sufficient time is needed to finish lighting all of the heaters before the temperature falls to critical levels.
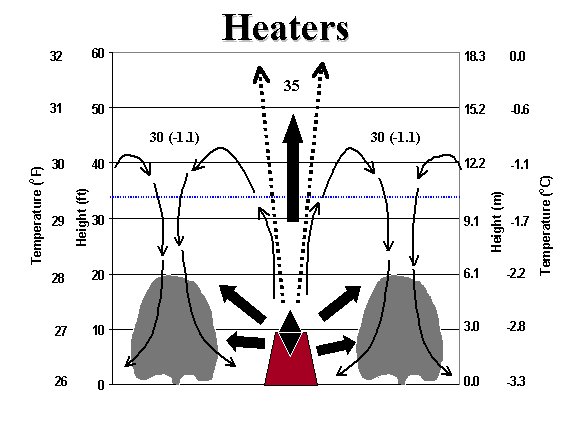
Figure 6. Schematic diagram showing the energy effect of a smudge pot heater on temperature within an inversion
PREDICTING MINIMUM TEMPERATURE
Predicting when the temperature falls to a critical value is important for starting active frost protection methods. In addition, the duration of temperature below the critical value is important for assessing potential damage. Starting at the proper temperature is important because it avoids losses resulting from starting too late and it saves energy by reducing the operation time of the various methods.
Assuming that the predicted minimum temperature (Tp) is correct and it will occur during the sunrise hour, then the nighttime hourly mean temperature trend from two hours after sunset through the sunrise hour is estimated using equations 1 and 2.
![]() (1)
(1)
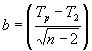 (2)
(2)
In Eq. 1, i =0 for the sunset hour and i=n for the sunrise hour the next morning. Ti is the mean air temperature for the ith hour and T2 is the mean temperature during the second hour following the sunset hour. Starting at hour i=2, which is two hours after the sunset hour, the mean hourly air temperature is predicted for each hour through the sunrise hour the next morning. The method is illustrated in Figure 3 (oF) and Figure 4 (oC). Note that the value for b depends on the temperature units (b=3.000 for oF and b=1.773 for oC). It is important to remember that this method will only work during radiation freezes with calm, clear nights when the temperature drops because of long wave radiation losses from the surface. Under windy and/or cloudy conditions, the temperature trend cannot be predicted using the approach presented here. However, radiation freezes do not occur during cloudy, windy conditions.
Generally, subtropical plants are most sensitive to freeze damage just before and during the harvest period when the temperature is lowest. The sensitivity of deciduous tree and vine crops to freezing temperature increases from first bloom to the small nut or fruit stages. Sensitivity is also higher when warm weather has preceded the frost night. Damage to a crop depends on how low the temperature goes and how long it is at the minimum temperature. Most published values for freeze damage to crops give the T10, T50, and T90 for 30 minutes at a given minimum air temperature. The T10, T50, and T90 are the air temperatures at which a 10%, 50% and 90% yield loss is expected when exposed to that temperature for 30 minutes. For example, given a T90=23oF=-5.0oC, we would expect more than 90% yield loss during the night of December 23, 1990 at the UC Lindcove Field Station.
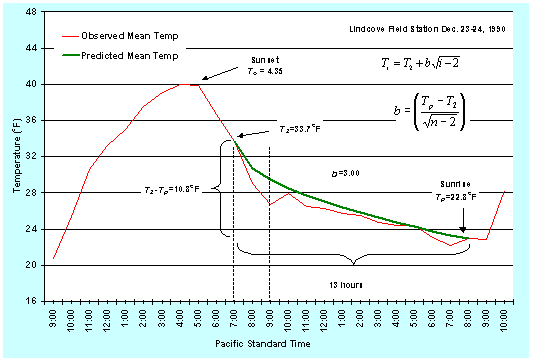
Figure 7. Observed and predicted hourly mean temperatures (oF) during a radiation freeze on December 23, 1990 at the University of California Lindcove Field Station.
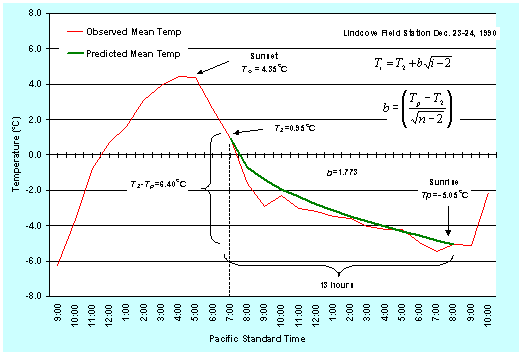
Figure 8. Observed and predicted hourly mean temperatures (oC) during a radiation freeze on December 23, 1990 at the University of California Lindcove Field Station.
At any given time during the night, the corresponding temperature is the predicted hourly mean temperature for the preceding hour. Therefore, the critical time for starting any active freeze-protection method is one hour before the critical damage temperature is expected. For example, using the 8:00 p.m. temperature in Figures 1 and 2, the temperature between 7:00 and 8:00 p.m. is predicted to average 30.5oF (-0.83oC). If unacceptable freeze damage is expected for half-an-hour exposure to a temperature of 30oF (-1.1oC), then, to be safe, the freeze protection should start at about 7:00 p.m. rather than at 8:00 p.m.
Note that this prediction method is fairly accurate, but it is not perfect. The observed temperatures between 7:00-10:00 p.m. in Figures 1 and 2 are less than the predicted temperatures. Figures 3 and 4 show the predicted versus observed temperatures over several days. Clearly, sometimes the predicted temperature is higher and sometimes it is lower than the observed. Damage could result if you rely only on the temperature prediction, so the model should be used as a guide to tell approximately when the temperature will reach a critical value. However, air temperature should be monitored during the night to accurately determine the time for starting freeze protection.
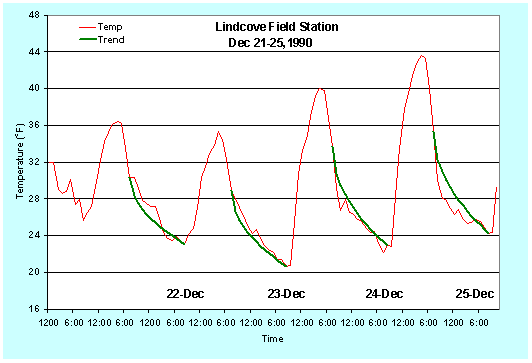
Figure 9. Predicted and observed temperature (oF) traces for December 21-25, 1990.
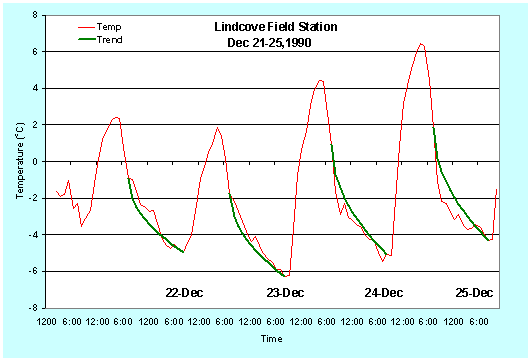
Figure 10. Predicted and observed temperature (oC) traces for December 21-25, 1990.
References
Allen, C.C. 1957. A simplified equation for minimum temperature prediction. Mon. Wea. Rev. 85, pp. 119-120.
Bagdonas, J.C., Georg, J.C., and Gerber, J.F. 1978. Techniques of frost prediction and methods of frost and cold protection. Technical Note No. 157. World Meteorol. Org. WMO No. 487. Geneva, Switzerland, pp. 1-160.
Crawford, T.T. 1965. Frost protection with wind machines and heaters. Meteorological Monographs 6:81-87.
Gerber, J.F. and J.D. Martsolf 1979. Sprinkling for frost and cold protection. In Modification of the aerial environment of crops. Eds. B.J. Barfield and J.F. Gerber. Amer. Soc. Of Civil Eng., St. Joseph, MO., pp. 327-333.
Hagood, L.B. 1967. An empirical method for forecasting radiation temperatures in the Lower Rio Grande Valley of Texas, Southern Regional Technical Memo. No. 33, National Weather Service.
Snyder, R.L. 1986. Frost protection: When to turn the sprinklers on and off. University of California One Sheet Answer 7165.
Young, F.D. 1920. Forecasting minimum temperatures in Oregon and California. Mon.Wea. Rev., 16, Supplement, pp. 53-60.